I have read the Privacy Policy
The collected data will be used exclusively to answer the request.
Electric double-layer capacitors are based on the operating principle of the electric double-layer that is formed at the interface between activated charcoal and an electrolyte.
The activated charcoal is used as an electrode, and the principle behind the capacitor is shown in Figure 1. Activated charcoal is used in its solid form, and the electrolytic fluid is liquid. When these materials come in contact with each other, the positive and negative poles are distributed relative to each other over an extremely short distance. Such a phenomenon is known as an electric double-layer. When an external electric field is applied, the electric double-layer that is formed in the vicinity of the activated charcoal's surface within the electrolytic fluid is used as the fundamental capacitor structure.
The electric double-layer design does not have the solid dielectric that is used in the previous designs, nor does it have the chemical reactions such as are found in batteries during charging and discharging. Rather this design has the following characteristics:
Taking advantage of the benefits described above, electric double-layer capacitors are used broadly in applications such as the following:
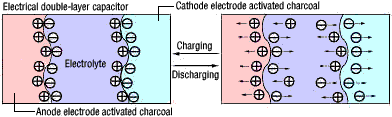
Figure 1: The Operating Principle in Electric Double-layer Capacitors
| Electric Double-layer Capacitor | Aluminum Electrolytic Capacitor | Ni-cd Battery | Lead Sealed Battery | |
|---|---|---|---|---|
| Use temperature range | −25 to 70°C | −55 to 125°C | −20 to 60°C | −40 to 60°C |
| Electrode materials | Activated charcoal | Aluminum | (+)NiOOH (-)Cd |
(+)PbO2 (-)Pb |
| Electrolytic fluid | Organic solvent | Organic solvent | KOH | H2SO4 |
| Method of electromotive force | Use of the naturally occurring electric double-layer effect as the dielectric. | Use of the aluminum oxide as the dielectric. | Use of chemical reaction. | Use of chemical reaction. |
| Pollution | None | None | Cd | Pb |
| Number of charge/discharge cycles | > 100,000 times | > 100,000 times | 500 times | 200 to 1000 times |
| Capacity per unit volume | 1 | 1/1000 | 100 | 100 |
Capacitance (Farad)
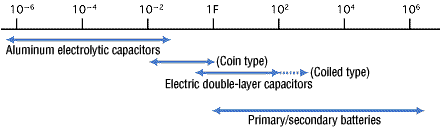
Figure 2